 
“These highly anticipated event-free survival results in this TNBC population build upon earlier findings from the KEYNOTE-522 trial and further support the potential use of Keytruda in these patients,” Vicki Goodman, M.D., vice president, clinical research, Merck Research Laboratories, said in a statement. Keytruda is currently approved under accelerated approval in the United States in combination with chemotherapy for the treatment of patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1. The trial is continuing to allow for additional follow-up of overall survival, a key secondary endpoint. In May, Merck had announced that KEYNOTE-522 met the dual primary endpoint of pathological complete response at the first interim analysis. No new safety concerns were seen in this fourth interim analysis. This is the first time an anti-PD-1/L1 therapy has demonstrated a statistically significant event-free survival result. Cortés J, Cescon DW, Rugo HS, et al: KEYNOTE-355: Final results from a randomized, double-blind phase III study of first-line pembrolizumab plus chemotherapy vs placebo plus chemotherapy for metastatic TNBC. After a median follow up of 39 months, the Keytruda regimen reduced the risk of event-free survival events by 37%. This study compared Keytruda and chemotherapy given before surgery followed by Keytruda (the Keytruda regimen) compared with adjuvant chemotherapy followed by placebo after surgery (the chemotherapy-placebo regimen). ESMO Trial Results Raise Possibility of Neoadjuvant Keytruda Use in Triple. KEYNOTE-355: Final results from a randomized, double-blind phase 3 study of first-line pembrolizumab + chemotherapy vs placebo + chemotherapy for metastatic TNBCīekijk hier het volledige overzicht van onze ESMO 2021 journaals en interviews.Merck’s Keytruda (pembrolizumab) demonstrated positive event-free survival (EFS) in patients with high-risk, early stage triple-negative breast cancer in the pivotal neoadjuvant/adjuvant phase 3 study KEYNOTE-522, according to results presented at European Society for Medical Oncology (ESMO) Virtual Plenary. Keynote-522 trial of Mercks Keytruda (pembrolizumab) in early TNBC. 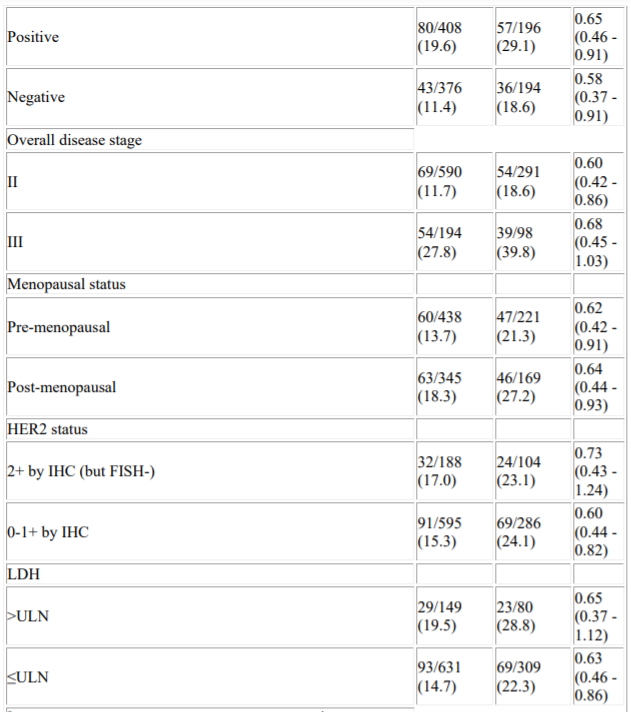
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
